Revolutionizing
Healthcare Through Better Insight
Founded in 2015 as a spin-off from UCLA's bioengineering program, PHASE Scientific Americas is a fast-growing biotechnology company dedicated to inspiring a new state of health. We are committed to providing innovative diagnostic and data tools needed to make better health decisions.
Our comprehensive product offerings are designed to address and fulfill a wide spectrum of market needs, making quality diagnostic testing more accessible to patients and providers.
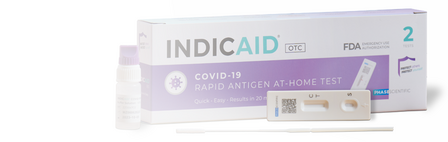
- INDICAID: OTC and POC rpaid antigen tests focused on the detection of common disease and conditions
- PHASiFY: Propietary sample preparation technology to advance molecular diagnostic testing
Our mission is clear: to redefine diagnostic testing through innovative technologies, PHASE Scientific Americas is at the forefront of transforming healthcare diagnostics.
Our Product & Services
Our Focus Areas
Cancer
Infectious Disease

Stay in the loop! Subscribe to our newsletter for the latest company updates.
Sign Up For Our Insights Newsletter TodayInspiringA New State of Health